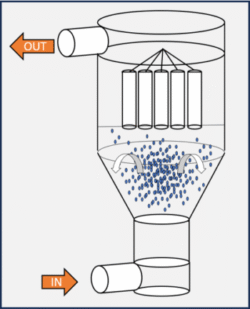
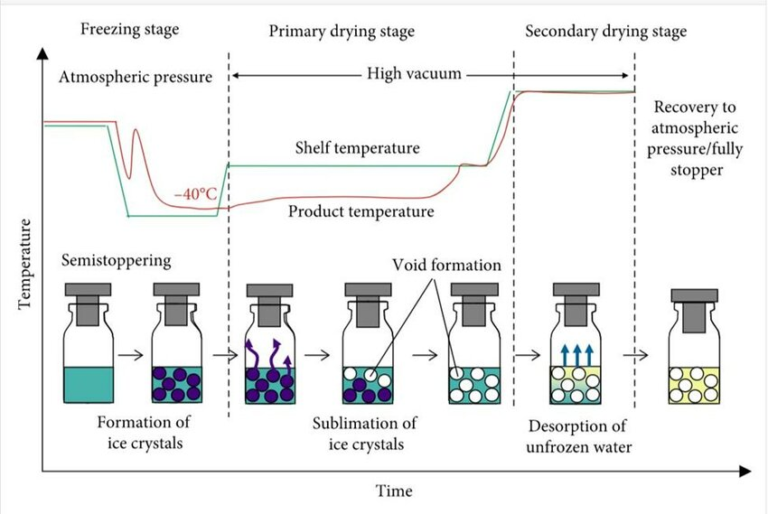
World of Injectables
Injectables in Pharmaceuticals: Mechanisms, Regulatory Pathways, Cost, Market Trends, and Innovations Across Therapeutic Domains Introduction Injectable pharmaceuticals have become a cornerstone of modern medicine, offering rapid, targeted, and often life-saving interventions across a spectrum of diseases. Their applications span general…